Our Advantage
With a science-first approach and a commitment to pharmaceutical European Good Manufacturing Practice (EU-GMP), we deliver standardized, high-purity formulations optimized for consistency, safety, and therapeutic impact. Our product range includes active pharmaceutical ingredients (APIs) and finished dosage forms designed to support modern evidence-based cannabis medicine.
Premium, Solventless Extracts
CRUDE OIL
Initial cannabis extract with all plant compounds
- Low purity, unrefined
- Unstable for formulation
- Contains unwanted plant lipids
- Inconsistent potency and effect
DISTILLATE OIL
Cannabinoid-rich extract refined through distillation
- Highly refined, stripped of minor cannabinoids and terpenes
- Lacks entourage effect
- Limited therapeutic versatility
- Common, low-margin commodity
CEYLON EXTRACTS
Extract made using hydrocarbons or heat/pressure only
- Full-spectrum, high-terpene extracts
- Preserves minor cannabinoids
- Enhanced therapeutic potential
- Supports premium pharma formulations
Full service, farm to pharmacy manufacturing
Leaders in product formulation.
A fully integrated platform for researching, formulating, manufacturing, and scaling cannabis-based preparations for commercial sale and clinical advancement.
Cultivation
Extraction
Purification
Formulation
Production
Packaging
From Extract to Medicine: Natural Products for Pharmaceutical Use

Resin

Rosin
Input Extracts

Capsules

Sublingual Oils

Galenic Forms

Inhalable
Finished Products
These dosage forms allow for consistent, scalable, and clinically relevant delivery of therapeutic cannabinoids.
From Extract to Medicine: Synthetic Products for Pharmaceutical Use
KT-182
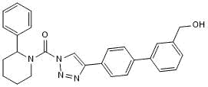
Brain-penetrant ABHD6 inhibitor at nanomolar potency for the treatment of seizures & epilepsy, neuropathic pain and migraines
ST-34 & ST-377

Brain-penetrant nanomolar potency for the treatment of GBM and melanoma brain metastasis
DX767
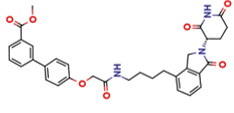
Membrane permeable PROTAC degrader of ABHD6 at nanomolar potency for the treatment of neuropathic pain and migraines
ST-360

Microtubule targeting nanomolar potency for the treatment of colorectal cancer
Broad Therapeutic Potential
Plant-Derived Molecules
Cannabidiol (CBD) + terpenes:
Improves sleep in people with insomnia (e.g., shift workers), as well as produces analgesic response for chronic pain.
Cannabigerol (CBG):
Treatment of anxiety & stress
Cannabinol (CBN):
Broad anti-inflammatory
Tetrahydrocannabinol (THC) + CBD:
Concerns about including THC, as it induces side effects. In combination with CBD, there are fewer side effects and possible therapeutic value as anti-inflammatory, including as skin ointment.
Synthetic Molecules
Medicinal chemistry optimization of fundamental cannabinoid scold - I.P. protected small molecules for the treatment of cancers, including glioblastoma, melanoma and colorectal cancer.
Biosimilars
Medicinal chemistry optimization of plant derived molecules - Develop novel I.P. around analogues of CBD, CBG, CBN and THC for the optimized treatment of sleep, chronic pain, inflammatory diseases, anxiety and stress.

